1.Introduction
The rate of a chemical reaction is the time taken for a given mass/amount of products to be formed. The rate of a chemical reaction is also the time taken for a given mass/amount of reactant to be consumed /used up.
Some reactions are too slow to be determined. e.g rusting ,decomposition of hydrogen peroxide and weathering.
Some reactions are too fast and instantaneous e.g. neutralization of acid and bases/alkalis in aqueous solution and double decomposition/precipitation.
Other reactions are explosive and very risky to carry out safely e.g. reaction of potassium with water and sodium with dilute acids.
The study of the rate of chemical reaction is useful in knowing the factors that influence the reaction so that efficiency and profitability is maximized in industries.
Theories of rates of reaction.
The rate of a chemical reaction is defined as the rate of change of concentration/amount of reactants in unit time. It is also the rate of formation of given concentration of products in unit time. i.e.
Rate of reaction = Change in concentration/amount of reactants
Time taken for the change to occur
Rate of reaction = Change in concentration/amount of products formed
Time taken for the products to form
For the above, therefore the rate of a chemical reaction is rate of decreasing reactants to form an increasing product.
The SI unit of time is second(s) but minutes and hours are also used.
(a)The collision theory
The collision theory is an application of the Kinetic Theory of matter which assumes matter is made up of small/tiny/minute particles like ions atoms and molecules.
Thecollision theory proposes that
(i)for a reaction to occur, reacting particles must collide.
(ii)not all collisions between reacting particles are successful in a reaction. Collisions that initiate a chemical reaction are called successful / fruitful/ effective collisions
(iii)the speed at which particles collide is called collision frequency.
The higher the collision frequency the higher the chances of successful / fruitful/ effective collisions to form products.
(iv)the higher the chances of successful collisions, the faster the reaction.
(v)the average distance between solid particles from one another is too big for them to meet and collide successfully.
(vi)dissolving substances in a solvent ,make the solvent a medium for the reaction to take place.
The solute particle distance is reduced as the particle ions are free to move in the solvent medium.
(vii)successful collisions take place if the particles colliding have the required energy and right orientation which increases their vibration and intensity of successful / fruitful/ effective collisions to form products.
(b)The Activation Energy(Ea) theory
The Enthalpy of activation(∆Ha) /Activation Energy(Ea) is the minimum amount of energy which the reactants must overcome before they react.Activation Energy(Ea) is usually required /needed in bond breaking of the reacting particles.
Bond breaking is an endothermic process that require an energy input.
The higher the bond energy the slower the reaction to start of.
Activation energy does not influence whether a reaction is exothermic or endothermic.
The energy level diagrams below shows the activation energy for exothermic and endothermic processes/reactions.
Energy level diagram showing the activation energy for exothermic processes /reactions.
The activated complex is a mixture of many intermediate possible products which may not exist under normal physical conditions ,but can theoretically exist.
Exothermic reaction proceeds without further heating /external energy because it generates its own energy/heat to overcome activation energy.
Endothermic reaction cannot proceed without further heating /external energy because it does not generates its own energy/heat to overcome activation energy. It generally therefore requires continuous supply of more energy/heat to sustain it to completion.
3. Measuring the rate of a chemical reaction.
The rate of a chemical reaction can be measure as:
(i)Volume of a gas in unit time;
– if reaction is producing a gas as one of the products.
– if reaction is using a gas as one reactants
(ii)Change in mass of reactants/products for solid products/reactants in unit time.
(iii)formation of a given mass of precipitate in unit time
(iv)a certain mass of reactants to completely form products/diminish.
Reactants may be homogenous or heterogenous.
-Homogenous reactions involve reactants in the same phase/state e.g. solid-solid,gas-gas,liquid-liquid.
-Heterogenous reactions involve reactants in the different phase/state e.g. solid-liquid,gas-liquid,solid-gas.
4. Factors influencing/altering/affecting/determining rate of reaction
The following factors alter/influence/affect/determine the rate of a chemical reaction:
(a)Concentration
(b)Pressure
(c) Temperature
(d)Surface area
(e)Catalyst
- Influence of concentration on rate of reaction
The higher the concentration, the higher the rate of a chemical reaction. An increase in concentration of the reactants reduces the distance between the reacting particles increasing their collision frequency to form products.
Practically an increase in concentration reduces the time taken for the reaction to take place.
Practical determination of effect of concentration on reaction rate
Method 1(a)
Reaction of sodium thisulphate with dilute hydrochloric acid
Procedure:
Measure 20cm3 of 0.05M sodium thisulphate into a 50cm3 glass beaker. Place the beaker on a white piece of filter paper with ink mark ‘X’ on it. Measure 20cm3 of 0.1M hydrochloric acid solution using a 50cm3 measuring cylinder. Put the acid into the beaker containing sodium thisulphate. Immediately start off the stop watch/clock. Determine the time taken for the ink mark ‘X’ to become invisible /obscured when viewed from above. Repeat the procedure by measuring different volumes of the acid and adding the volumes of the distilled water to complete table 1. Sample results:Table 1.
| Volume of acid(cm3) | Volume of water(cm3) | Volume of sodium thiosulphate(cm3) | Time taken for mark ‘X’ to be invisible/obscured(seconds) | Reciprocal of time 1 t |
| 20.0 | 0.0 | 20.0 | 20.0 | 5.0 x 10-2 |
| 18.0 | 2.0 | 20.0 | 23.0 | 4.35 x 10-2 |
| 16.0 | 4.0 | 20.0 | 27.0 | 3.7 x 10-2 |
| 14.0 | 6.0 | 20.0 | 32.0 | 3.13 x 10-2 |
| 12.0 | 8.0 | 20.0 | 42.0 | 2.38 x 10-2 |
| 10.0 | 10.0 | 20.0 | 56.0 | 1.78 x 10-2 |
For most examining bodies/councils/boards the above results score for:
(a) complete table as evidence for all the practical work done and completed.
(b) (i)Consistent use of a decimal point on time as evidence of understanding/knowledge of the degree of accuracy of stop watches/clock.
(ii)Consistent use of a minimum of four decimal points on inverse/reciprocal of time as evidence of understanding/knowledge of the degree of accuracy of scientific calculator.
(c) accuracy against a school value based on candidate’s teachers-results submitted.
(d) correct trend (time increase as more water is added/acid is diluted) in conformity with expected theoretical results.
Sample questions
1. On separate graph papers plot a graph of:
(i)volume of acid used(x-axis) against time. Label this graph I
(ii) volume of acid used(x-axis) against 1/t. Label this graph II
2. Explain the shape of graph I
Diluting/adding water is causes a decrease in concentration.
Decrease in concentration reduces the rate of reaction by increasing the time taken for reacting particle to collide to form products.
Sketch sample Graph I
Method 1(b)
Reaction of sodium thisulphate with dilute hydrochloric acid
You are provided with
2.0M Hydrochloric acid
0.4M sodium thiosulphate solution
Procedure:
Measure 10cm3 of sodium thisulphate into a 50cm3 glass beaker. Place the beaker on a white piece of filter paper with ink mark ‘X’ on it.
Add 5.0cm3 of hydrochloric acid solution using a 10cm3 measuring cylinder into the beaker containing sodium thisulphate.
Immediately start off the stop watch/clock. Determine the time taken for the ink mark ‘X’ to become invisible /obscured when viewed from above.
Repeat the procedure by measuring different volumes of the thiosulphate and adding the volumes of the distilled water to complete table 1.
Sample results:Table 1.
| Volume of acid(cm3) | Volume of water (cm3) | Volume of sodium thiosulphate (cm3) | Concentation of sodium thisulphate in molesdm-3 | Time(T) taken for mark ‘X’ to be invisible/ obscured(seconds) | T-1 |
| 5.0 | 0.0 | 25.0 | 0.4 | 20.0 | 5.0 x 10-2 |
| 5.0 | 5.0 | 20.0 | 0.32 | 23.0 | 4.35 x 10-2 |
| 5.0 | 10.0 | 15.0 | 0.24 | 27.0 | 3.7 x 10-2 |
| 5.0 | 15.0 | 10.0 | 0.16 | 32.0 | 3.13 x 10-2 |
Note concentration of diluted solution is got:
C1V1=C2V2 => 0.4 x 25 = C2x 25 =0.4M
C1V1=C2V2 => 0.4 x 20 = C2x 25 =0.32M
C1V1=C2V2 => 0.4 x 15 = C2x 25 =0.24M
C1V1=C2V2 => 0.4 x 10 = C2x 25 =0.16M
Sample questions
1. On separate graph papers plot a graph of:
(i)Concentration of sodium thiosulphate against time. Label this graph I
(ii)Concentration of sodium thiosulphate against against T-1.Label this graph II
2. Explain the shape of graph I
Diluting/adding water causes a decrease in concentration.
Decrease in concentration reduces the rate of reaction by increasing the time taken for reacting particle to collide to form products.
From graph II
Determine the time taken if
(i)12cm3 of sodium thisulphate is diluted with 13cm3 of water.
At 12cm3 concentration of sodium thisulphate
= C1V1=C2V2 => 0.4 x 1 2 = C2x 25 =0.192M
From correct graph at concentration 0.192M => 2.4 x10-2
I/t = 2.4 x10-2 t = 41.6667seconds
(ii)22cm3 of sodium thisulphate is diluted with 3cm3 of water.
At 22cm3 concentration of sodium thisulphate
= C1V1=C2V2 => 0.4 x 22 = C2x 25 =0.352M
From correct graph at concentration 0.352M => 3.6 x10-2
I/t = 3.6 x10-2 t = 27.7778seconds
Determine the volume of water and sodium thiosulphate if T-1 is 3.0 x10-1
From correct graph at T-1 = 3.0 x10-1 => concentration = 0.65 M
= C1V1=C2V2 => 0.4 x 25 = 0.65 M x V2 = 15.3846cm3
Volume of water = 25 – 15.3846cm3 = 9.6154cm3
Determine the concentration of hydrochloric acid if 12cm3 of sodium thiosulphate and 13cm3 of water was used.
At 12cm3 concentration of sodium thisulphate
= C1V1=C2V2 => 0.4 x 1 2 = C2x 25 =0.192M
Mole ratio Na2S2 O3 :HCl =1:2
Moles of Na2S2 O3 = 0.192M x 12 => 2.304 x 10-3 moles
1000
Mole ratio HCl =2.304 x 10-1 moles = 1.152 x 10-3 moles
2
Molarity o f HCl = 1.152 x 10-3 moles x 1000 = 0.2304M
5.0
Method 2
Reaction of Magnesium with dilute hydrochloric acid
Procedure
Scub 10centimeter length of magnesium ribbon with sand paper/steel wool. Measure 40cm3 of 0.5M dilute hydrochloric acid into a flask .Fill a graduated gas jar with water and invert it into a trough. Stopper the flask and set up the apparatus to collect the gas produced as in the set up below:
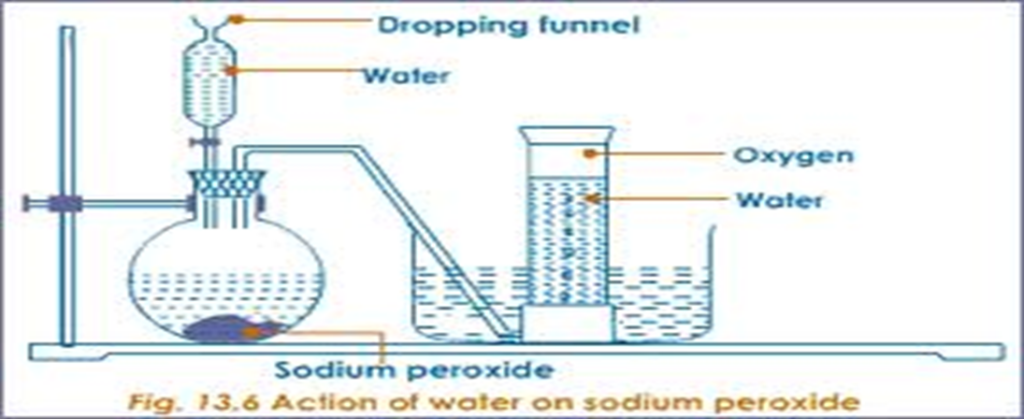
Carefully remove the stopper, carefully put the magnesium ribbon into the flask . cork tightly. Add the acid into the flask. Connect the delivery tube into the gas jar. Immediately start off the stop watch and determine the volume of the gas produced after every 30 seconds to complete table II below.
Sample results: Table II
| Time(seconds) | 0 | 30 | 60 | 90 | 120 | 150 | 180 | 210 | 240 |
| Volume of gas produced(cm3) | 0.0 | 20.0 | 40.0 | 60.0 | 80.0 | 90.0 | 95.0 | 96.0 | 96.0 |
Sample practice questions
1.Plot a graph of volume of gas produced (y-axis) against time
2.Explain the shape of the graph.
The rate of reaction is faster when the concentration of the acid is high .
As time goes on, the concentration of the acid decreases and therefore less gas is produced.
When all the acid has reacted, no more gas is produced after 210 seconds and the graph flattens.
3.Calculate the rate of reaction at 120 seconds
From a tangent at 120 seconds rate of reaction = Change in volume of gas
Change in time
=> From the tangent at 120seconds V2 – V1 = 96-84 = 12 = 0.2cm3sec-1
T2 – T1 150-90 60
4. Write an ionic equation for the reaction taking place.
Mg2+(s) + 2H+(aq) -> Mg2+(aq) + H2 (g)
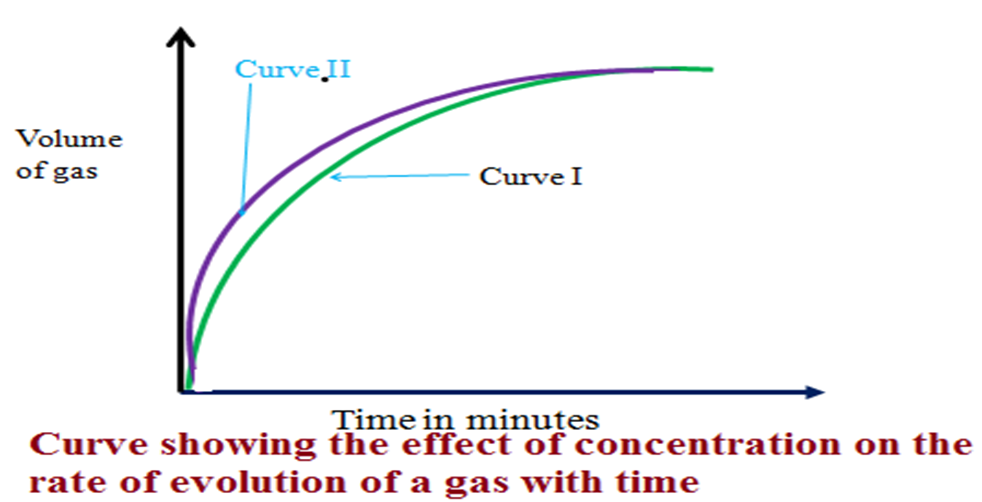
5. On the same axis sketch then explain the curve that would be obtained if:
(i) 0.1 M hydrochloric acid is used –Label this curve I
(ii)1.0 M hydrochloric acid is used –Label this curve II
Observation:
Curve I is to the right
Curve II is to the left
Explanation
A decrease in concentration shift the rate of reaction graph to the right as more time is taken for completion of the reaction.
An increase in concentration shift the rate of reaction graph to the left as less time is taken for completion of the reaction.
Both graphs flatten after some time indicating the completion of the reaction.
b)Influence of pressure on rate of reaction
Pressure affects only gaseous reactants.
An increase in pressure reduces the volume(Boyles law) in which the particles are contained.
Decrease in volume of the container bring the reacting particles closer to each other which increases their chances of effective/successful/fruitful collision to form products.
An increase in pressure therefore increases the rate of reaction by reducing the time for reacting particles of gases to react.
At industrial level, the following are some reactions that are affected by pressure:
(a)Haber process for manufacture of ammonia
N2(g) + 3H2(g) -> 2NH3(g)
(b)Contact process for manufacture of sulphuric(VI)acid
2SO2(g) + O2(g) -> 2SO3(g)
(c)Ostwalds process for the manufacture of nitric(V)acid
4NH3(g) + 5O2(g) -> 4NO (g) + 6H2O (l)
The influence of pressure on reaction rate is not felt in solids and liquids.
This is because the solid and liquid particles have fixed positions in their strong bonds and therefore no degree of freedom (Kinetic Theory of matter)
c)Influence of temperature on rate of reaction
An increase in temperature increases the kinetic energy of the reacting particles by increasing their collision frequency.
Increase in temperature increases the particles which can overcome the activation energy (Ea).
A 10oC rise in temperature doubles the rate of reaction by reducing the time taken for the reaction to complete by a half.
Practical determination of effect of Temperature on reaction rate
Method 1
Reaction of sodium thisulphate with dilute hydrochloric acid
Procedure:
Measure 20cm3 of 0.05M sodium thisulphate into a 50cm3 glass beaker.
Place the beaker on a white piece of filter paper with ink mark ‘X’ on it.
Determine and record its temperature as room temperature in table 2 below.
Measure 20cm3 of 0.1M hydrochloric acid solution using a 50cm3 measuring cylinder.
Put the acid into the beaker containing sodium thisulphate.
Immediately start off the stop watch/clock.
Determine the time taken for the ink mark ‘X’ to become invisible /obscured when viewed from above.
Measure another 20cm3 separate portion of the thisulphate into a beaker, heat the solution to 30oC.
Add the acid into the beaker and repeat the procedure above. Complete table 2 below using different temperatures of the thiosulphate.
Sample results:Table 2.
| Temperature of Na2S2O3 | Room temperature | 30 | 40 | 50 | 60 |
| Time taken for mark X to be obscured /invisible (seconds) | 50.0 | 40.0 | 20.0 | 15.0 | 10.0 |
| Reciprocal of time(1/t) | 0.02 | 0.025 | 0.05 | 0.0667 | 0.1 |
Sample practice questions
Method 2
Reaction of Magnesium with dilute hydrochloric acid
Procedure
Scub 5centimeter length of magnesium ribbon with sand paper/steel wool.
Cut the piece into five equal one centimeter smaller pieces.
Measure 20cm3 of 1.0M dilute hydrochloric acid into a glass beaker .
Put one piece of the magnesium ribbon into the acid, swirl.
Immediately start off the stop watch/clock.
Determine the time taken for the effervescence/fizzing/bubbling to stop when viewed from above.
Record the time in table 2 at room temperature.
Measure another 20cm3 portions of 1.0M dilute hydrochloric acid into a clean beaker.
Heat separately one portion to 30oC, 40oC , 50oC and 60oC and adding 1cm length of the ribbon and determine the time taken for effervescence /fizzing /bubbling to stop when viewed from above .
Record each time to complete table 2 below using different temperatures of the acid.
Sample results:Table 1.
| Temperature of acid(oC) | Room temperature | 30 | 40 | 50 | 60 |
| Time taken effervescence to stop (seconds) | 80.0 | 50.0 | 21.0 | 13.5 | 10.0 |
| Reciprocal of time(1/t) | 0.0125 | 0.02 | 0.0476 | 0.0741 | 0.1 |
2.(a)Calculate the number of moles of magnesium used given that 1cm of magnesium has a mass of 1g.(Mg= 24.0)
Moles = Mass of magnesium => 1.0 = 4.167 x 10 -2 moles
Molar mass of Mg 24
(b)Calculate the number of moles of hydrochloric acid used
Moles of acid = molarity x volume of acid
1000
=> 1.0 x 20 = 2.0 x 10 -2 moles
1000
(c)Calculate the mass of magnesium that remain unreacted
Mole ratio Mg: HCl = 1:2
Moles Mg = ½ moles HCl
=> ½ x 2.0 x 10 -2 moles = 1.0 x 10 -2 moles
Mass of reacted Mg = moles x molar mass
=> 1.0 x 10 -2 moles x 24 = 0.24 g
Mass of unreacted Mg = Original total mass – Mass of reacted Mg
=> 1.0 g – 0.24 = 0.76 g
(b)Calculate the total volume of hydrogen gas produced during the above reactions.
Mole ratio Mg : H2 = 1:1
Moles of Mg that reacted per experiment = moles H2 =1.0 x 10 -2 moles
Volume of Hydrogen at s.t.p produced per experiment = moles x 24 dm3
=> 1.0 x 10 -2 moles x 24 dm3 = 0.24dm3
Volume of Hydrogen at s.t.p produced in 5 experiments =0.24 dm3 x 5
= 1.2 dm3
3.(a)At what temperature was the time taken for magnesium to react equal to:
(i)70seconds
70 seconds => 1/t =1/70 =0.01429
Reading directly from a correctly plotted graph 0.01429 => 28.0 oC
(ii)40seconds
40 seconds => 1/t =1/40 =0.025
Reading directly from a correctly plotted graph 0.025 => 32.0 oC
(b)What is the time taken for magnesium to react if the reaction was done at:
(i) 55.0 oC
Reading directly from a correctly plotted graph at 55.0 oC=> 1/t = 8.0 x 10-2
=> t = 1/8.0 x 10-2 = 12.5 seconds
(ii) 47.0 oC
Reading directly from a correctly plotted graph at 47.0 oC=> 1/t = 6.0 x 10-2
=> t = 1/6.0 x 10-2 = 16.6667 seconds
(iii) 33.0 oC
Reading directly from a correctly plotted graph at 33.0 oC=> 1/t = 2.7 x 10-2
=> t = 1/2.7 x 10-2 = 37.037 seconds
4. Explain the shape of the graph.
Increase in temperature increases the rate of reaction as particles gain kinetic energy increasing their frequency and intensity of collision to form products.
d)Influence of surface area on rate of reaction
Surface area is the area of contact. An increase in surface area is a decrease in particle size. Practically an increase in surface area involves chopping /cutting solid lumps into smaller pieces/chips then crushing the chips into powder. Chips thus have a higher surface area than solid lumps but powder has a highest surface area.
An increase in surface area of solids increases the area of contact with a liquid solution increasing the chances of successful/effective/fruitful collision to form products. The influence of surface area on rate of reaction is mainly in heterogeneous reactions.
Reaction of chalk/calcium carbonate on dilute hydrochloric acid
Procedure
Measure 20cm3 of 1.0 M hydrochloric acid into three separate conical flasks labeled C1 C2 and C3 .
Using a watch glass weigh three separate 2.5g a piece of white chalk. Place the conical flask C1 on an electronic balance.
Reset the balance scale to 0.0.
Put one weighed sample of the chalk into the acid in the conical flask. Determine the scale reading and record it at time =0.0.
Simultaneously start of the stop watch.
Determine and record the scale reading after every 30 seconds to complete Table I .Repeat all the above procedure separately with C2 and C3 to complete Table II and Table III by cutting the chalk into small pieces/chips for C2 and crushing the chalk to powder for C3
Sample results:Table 1.
| Time(seconds) | 0.0 | 30.0 | 60.0 | 90.0 | 120.0 | 150.0 | 180.0 | 210.0 | 240.0 |
| Mass of CaCO3 | 2.5 | 2.0 | 1.8 | 1.4 | 1.2 | 1.0 | 0.8 | 0.5 | 0.5 |
| Loss in mass | 0.0 | 0.5 | 0.7 | 1.1 | 1.3 | 1.5 | 1.7 | 2.0 | 2.0 |
Sample results:Table 1I.
| Time(seconds) | 0.0 | 30.0 | 60.0 | 90.0 | 120.0 | 150.0 | 180.0 | 210.0 | 240.0 |
| Mass of CaCO3 | 2.5 | 1.9 | 1.5 | 1.3 | 1.0 | 0.8 | 0.5 | 0.5 | 0.5 |
| Loss in mass | 0.0 | 0.6 | 1.0 | 1.2 | 1.5 | 1.7 | 2.0 | 2.0 | 2.0 |
Sample results:Table III.
| Time(seconds) | 0.0 | 30.0 | 60.0 | 90.0 | 120.0 | 150.0 | 180.0 | 210.0 | 240.0 |
| Mass of CaCO3 | 2.5 | 1.8 | 1.4 | 1.0 | 0.8 | 0.5 | 0.5 | 0.5 | 0.5 |
| Loss in mass | 0.0 | 0.7 | 1.1 | 1.5 | 1.7 | 2.0 | 2.0 | 2.0 | 2.0 |
Sample questions:
1.Calculate the loss in mass made at the end of each time from the original to complete table I,II and III
2.On the same axes plot a graph of total loss in mass against time (x-axes) and label them curve I, II, and III from Table I, II, and III.
3.Explain why there is a loss in mass in all experiments.
Calcium carbonate react with the acid to form carbon(IV)oxide gas that escape to the atmosphere.
4.Write an ionic equation for the reaction that take place
CaCO3(s) + 2H+(aq) -> Ca2+(aq) + H2O(l) + CO2(g)
5.Sulphuric(VI)acid cannot be used in the above reaction. On the same axes sketch the curve which would be obtained if the reaction was attempted by reacting a piece of a lump of chalk with 0.5M sulphuric(VI)acid. Label it curve IV. Explain the shape of curve IV.
Calcium carbonate would react with dilute 0.5M sulphuric(VI)acid to form insoluble calcium sulphate(VI) that coat /cover unreacted Calcium carbonate stopping the reaction from reaching completion.
6.Calculate the volume of carbon(IV)oxide evolved(molar gas volume at room temperature = 24 dm3, C= 12.0, O= 16.O Ca=40.0)
Method I
Mole ratio CaCO3(s) : CO2(g) = 1:1
Moles CaCO3(s) used = Mass CaCO3(s) = 0.025 moles
Molar mass CaCO3(s)
Moles CO2(g) = 0.025 moles
Volume of CO2(g) = moles x molar gas volume
=>0.025 moles x 24 dm3 = 0.600 dm3/600cm3
Method II
Molar mass of CaCO3(s) = 100g produce 24 dm3 of CO2(g)
Mass of CaCO3(s) =2.5 g produce 2.5 x 24 = 0.600dm3
100
7.From curve I ,determine the rate of reaction (loss in mass per second)at time 180 seconds on the curve.
From tangent at 180 seconds on curve I
Rate = M2-M1 => 2.08 – 1.375 = 0.625 = 0.006944g sec-1
T2– T1 222-132 90
8.What is the effect of particle size on the rate of reaction?
A larger surface area is a reduction in particle size which increases the area of contact between reacting particles increasing their collision frequency.
Theoretical examples
1. Excess marble chips were put in a beaker containing 100cm3 of 0.2M hydrochloric acid. The beaker was then placed on a balance and total loss in mass recorded after every two minutes as in the table below.
| Time(minutes) | 0.0 | 2.0 | 4.0 | 6.0 | 8.0 | 10.0 | 12.0 |
| Loss in mass(g) | 0.0 | 1.80 | 2.45 | 2.95 | 3.20 | 3.25 | 3.25 |
(a)Why was there a loss in mass?
Carbon (IV) oxide gas was produced that escape to the surrounding
(b)Calculate the average rate of loss in mass between:
(i) 0 to 2 minutes
Average rate =M2-M1 => 1.80 – 0.0 = 1.8 = 9.00g min-1
T2– T1 2.0 – 0.0 2
(i) 6 to 8 minutes
Average rate =M2-M1 => 3.20 – 2.95 = 0.25 = 0.125g min-1
T2– T1 8.0 – 6.0 2
(iii) Explain the difference between the average rates of reaction in (i) and(ii) above.
Between 0 and 2 minutes , the concentration of marble chips and hydrochloric acid is high therefore there is a higher collision frequency between the reacting particles leading to high successful rate of formation of products.
Between 6 and 8 minutes , the concentration of marble chips and hydrochloric acid is low therefore there is low collision frequency between the reacting particles leading to less successful rate of formation of products.
(c)Write the equation for the reaction that takes place.
CaCO3(s) + 2HCl (aq) -> CaCO3 (aq) + H2O(l) + CO2(g)
(d)State and explain three ways in which the rate of reaction could be increased.
(i)Heating the acid- increasing the temperature of the reacting particles increases their kinetic energy and thus collision frequency.
(ii)Increasing the concentration of the acid-increasing in concentration reduces the distances between the reacting particles increasing their chances of effective/fruitful/successful collision to form products faster.
(iii)Crushing the marble chips to powder-this reduces the particle size/increase surface area increasing the area of contact between reacting particles.
(e)If the solution in the beaker was evaporated to dryness then left overnight in the open, explain what would happen.
It becomes wet because calcium (II) chloride absorbs water from the atmosphere and form solution/is deliquescent.
(f)When sodium sulphate (VI) was added to a portion of the contents in the beaker after the reaction , a white precipitate was formed .
(i)Name the white precipitate.
Calcium(II)sulphate(VI)
(ii)Write an ionic equation for the formation of the white precipitate
Ca2+(aq) + SO42-(aq)->CaSO4(s)
(iii)State one use of the white precipitate
-Making plaster for building
-Manufacture of plaster of Paris
-Making sulphuric(VI)acid
(g)(i) Plot a graph of total loss in mass(y-axes) against time
(ii)From the graph, determine the rate of reaction at time 2 minutes.
From a tangent/slope at 2 minutes;
Rate of reaction = Average rate =M2-M1 => 2.25 – 1.30 = 0.95 = 0.3958g min-1
T2– T1 3.20 – 0.8 2.4
(iii)Sketch on the same axes the graph that would be obtained if 0.02M hydrochloric acid was used. Label it curve II
e) Influence of catalyst on rate of reaction
Catalyst is a substance that alter the rate /speed of a chemical reaction but remain chemically unchanged at the end of a reaction. Biological catalysts are called enzymes. A catalyst does not alter the amount of products formed but itself may be altered physically e.g. from solid to powder to fine powder. Like biological enzymes, a catalyst only catalyse specific type of reactions
Most industrial catalysts are transition metals or their compounds. Catalyst works by lowering the Enthalpy of activation(∆Ha)/activation energy (Ea) of the reactants .The catalyst lowers the Enthalpy of activation(∆Ha)/activation energy (Ea) by:
(i) forming short lived intermediate compounds called activated complex that break up to form the final product/s
(ii) being absorbed by the reactants thus providing the surface area on which reaction occurs.
A catalyst has no effect on the enthalpy of reaction ∆Hr but only lowers the Enthalpy of activation(∆Ha)/activation energy (Ea)It thus do not affect/influence whether the reaction is exothermic or endothermic as shown in the energy level diagrams below.
Energy level diagram showing the activation energy for exothermic processes /reactions.
The following are some catalysed reaction processes.
(a)The contact process
Vanadium(V) Oxide(V2O5) or platinum(Pt) catalyses the oxidation of sulphur(IV)oxide during the manufacture of sulphuric(VI) acid from contact process.
SO2(g) + O2(g) —-V2O5–> SO3(g)
To reduce industrial cost of manufacture of sulphuric (VI) acid from contact process Vanadium(V) Oxide(V2O5) is used because it is cheaper though it is easily poisoned by impurities.
(b)Ostwalds process
Platinum promoted with Rhodium catalyses the oxidation of ammonia to nitrogen(II)oxide and water during the manufacture of nitric(V)acid
4NH3(g) + 5O2(g) —-Pt/Rh–> 4NO (g) + 6H2O(l)
(c)Haber process
Platinum or iron catalyses the combination of nitrogen and hydrogen to form ammonia gas
N2(g) + 3H2(g) —Pt or Fe—> 2NH3(g)
(d)Hydrogenation/Hardening of oil to fat
Nickel (Ni) catalyses the hydrogenation of unsaturated compound containing – C=C- or –C=C- to saturated compounds without double or triple bond
This process is used is used in hardening oil to fat.
(e)Decomposition of hydrogen peroxide
Manganese(IV)oxide speeds up the rate of decomposition of hydrogen peroxide to water and oxygen gas.
This process/reaction is used in the school laboratory preparation of Oxygen.
2H2O2 (g) —-MnO2–> O2(g) + 2H2O(l)
(f)Reaction of metals with dilute sulphuric(VI)acid
Copper(II)sulphate(VI) speeds up the rate of production of hydrogen gas from the reaction of Zinc and dilute sulphuric(VI)acid.
This process/reaction is used in the school laboratory preparation of Hydrogen.
H2 SO4 (aq) + Zn(s) —-CuSO4–> ZnSO4 (aq) + H2(g)
(g) Substitution reactions
When placed in bright sunlight or U.V /ultraviolet light , a mixture of a halogen and an alkane undergo substitution reactions explosively to form halogenoalkanes. When paced in diffused sunlight the reaction is very slow.
e.g. CH4(g) + Cl2(g) —u.v. light–> CH3Cl(g) + HCl(g)
(h)Photosynthesis
Plants convert carbon(IV)oxide gas from the atmosphere and water from the soil to form glucose and oxygen as a byproduct using sunlight / ultraviolet light.
6CO2(g) + 6H2O(l) —u.v. light–> C6H12O6(g) + O2(g)
(i)Photography
Photographic film contains silver bromide emulsion which decomposes to silver and bromine on exposure to sunlight.
2AgBr(s) —u.v/sun light–> 2Ag(s) + Br2(l)
When developed, the silver deposits give the picture of the object whose photograph was taken depending on intensity of light. A picture photographed in diffused light is therefore blurred.
Practical determination of effect of catalyst on decomposition of hydrogen peroxide
Measure 5cm3 of 20 volume hydrogen peroxide and then dilute to make 40cm3 in a measuring cylinder by adding distilled water.
Divide it into two equal portions.
(i)Transfer one 20cm3volume hydrogen peroxide into a conical/round bottomed/flat bottomed flask. Cork and swirl for 2 minutes. Remove the cork. Test the gas produced using a glowing splint. Clean the conical/round bottomed/flat bottomed flask.
(ii)Put 2.0g of Manganese (IV) oxide into the clean conical/round bottomed/flat bottomed flask. Stopper the flask.
Transfer the second portion of the 20cm3volume hydrogen peroxide into a conical/round bottomed/flat bottomed flask through the dropping/thistle funnel. Connect the delivery tube to a calibrated/graduated gas jar as in the set up below.
Start off the stop watch and determine the volume of gas in the calibrated/graduated gas jar after every 30 seconds to complete Table 1.
(iii)Weigh a filter paper .Use the filter paper to filter the contents of the conical conical/round bottomed/flat bottomed flask. Put the residue on a sand bath to dry. Weigh the dry filter paper again .Determine the new mass Manganese (IV) oxide.
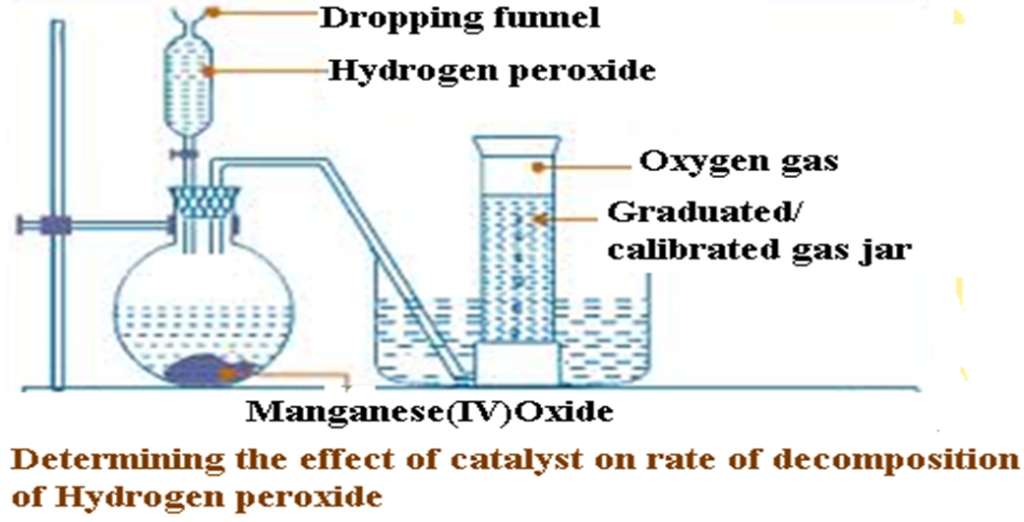
| Time(seconds) | 0.0 | 30.0 | 60.0 | 90.0 | 120.0 | 150.0 | 180.0 | 210.0 | 240.0 | 270.0 |
| Volume of gas (cm3) | 0.0 | 20.0 | 40.0 | 60.0 | 80.0 | 90.0 | 95.0 | 96.0 | 96.0 | 96.0 |
| Mass of MnO2 before reaction(g) | Mass of MnO2 after reaction(g) |
| 2.0 | 2.0 |
Plot a graph of volume of gas produced against time(x-axes)
b) On the same axes, plot a graph of the uncatalysed reaction.
(c) Explain the changes in mass of manganese(IV)oxide before and after the reaction.
The mass of MnO2 before and after the reaction is the same but a more fine powder after the experiment. A catalyst therefore remains unchanged chemically but may physically change.
